Allergy Blog
Updates to Adrenaline Auto-Injectors
Emerade – Bausch & Lomb
Ireland
The HPRA (Health Products Regulatory Authority) in Ireland announced that PharmaSwiss was recalling all lots (batches) of Emerade 150mcg pre-filled pens, Emerade 300mcg pre-filled pens and Emerade 500mcg pre-filled pens.
Anyone who possesses an Emerade pen for their own use or for someone in their care is urged to ensure that their pens are brought back to their pharmacy and immediately replaced with an alternative product. The HPRA states that no other brand of adrenaline pens is impacted by this recall.
See the full recall notice here: https://www.hpra.ie/homepage/medicines/news-events/item?t=/patient-level-recall-of-all-batches-of-emerade-adrenaline-auto-injector-pens&id=48720c26-9782-6eee-9b55-ff00008c97d0
United Kingdom
The MHRA has now recalled all batches of Emerade 150, 300 and 500 microgram auto-injectors. In the UK there are two alternative adrenaline auto-injector devices available – Jext and EpiPen. However, the different brands of adrenaline auto-injectors are not used in exactly the same way and therefore specific training and advice is required for each of the devices. The Medicines and Healthcare products Regulatory Agency (MHRA) said healthcare professionals were being asked to contact patients who use Emerade pens. Both patients and carers should return all Emerade auto-injectors to their local pharmacy once they have obtained a prescription for, and been supplied with, an alternative brand, said the MHRA.
See the full drug alert notice here: https://www.gov.uk/drug-device-alerts/class-2-medicines-recall-emerade-500-micrograms-solution-for-injection-in-pre-filled-syringe-pl-33616-0015-el-20-a-23
Information on Emerade auto-injector recalls also available from https://www.anaphylaxis.org.uk/tag/emerade/?fbclid=IwAR3YMQ9rMmdfQufRGVOdQVoxU2T58W8gWntMiszUa7VbYG1FbFg-EJmzut8
EpiPen – Mylan
United Kingdom
Supply has stabilised further in the UK and there is good availability of both EpiPen Jr 0.15mg and EpiPen 0.3mg AAIs
Full information at the link https://www.anaphylaxis.org.uk/wp-content/uploads/2020/04/EpiPen-Supply-UK-Website-Update_23.04-1.pdf
There is an extended use for specific lot numbers of EpiPen® 0.3mg Adrenaline Auto‐injectors only.
This letter is sent in agreement with the UK medicines regulator, Medicines and Healthcare Products Regulatory Agency (MHRA), to inform you of the following: Mylan UK have obtained acceptance from the MHRA to extend the use of specific lot numbers of EpiPen® 0.3mg auto‐injectors, by 4 months beyond the labelled expiry date.
This batch-specific extended use does not apply to EpiPen® 0.15mg AAI or any lot number of EpiPen 0.3 mg auto-injectors not specified.
It is important that patients must continue to adhere to the labelled expiry date on any EpiPen® not covered by the lot numbers below.
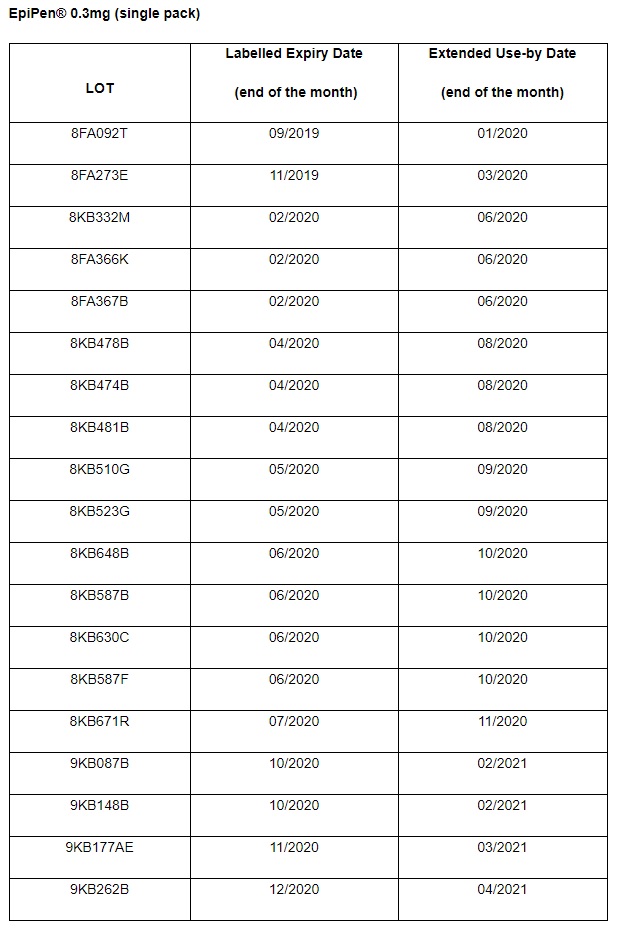
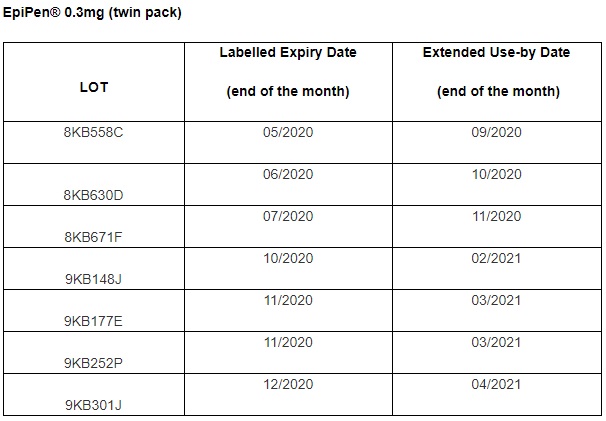
Find more information here: https://www.bsaci.org/announcements/epipen-0-3mg-extended-use-beyond-labelled-expiry-date-for-specific-lots
Disclaimer: The information provided is for informational purposes only and is not intended to be a substitute for professional medical advice, diagnosis or treatment. Allergy Lifestyle Limited (t/a) Allergy Lifestyle) uses reasonable endeavours to check the accuracy of information provided however no warranty is given that they are error-free.


