Allergy Blog
New Adrenaline Auto Injector – Symjepi
Adamis Pharmaceuticals have just received FDA approval for their new prefilled epinephrine syringe, Symjepi. The syringe contains 0.3 mg of epinephrine. Adamis Pharmaceuticals are currently seeking approval for a 0.15 mg syringe also.
The price of the Symjepi syringe has not yet been set but Adamis have said that it will be cheaper than the EpiPen® epinephrine auto-injector. It should be on shelves in the US later this year.
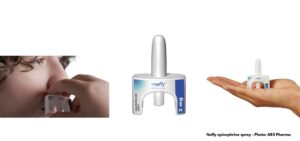
Maker’s Adamis say that the Symjepi injector is smaller than the EpiPen® and easier to use.
The new injector could take market share away from Mylan’s EpiPen® injectors. The company came under fire last year for the price of their auto-injector which had increased nearly 500% in price over the course of 7 years. The company lost more than 25% of its share in the market since the beginning of 2016.
How it’s used
Symjepi is a single use prefilled syringe and cannot be reused. This syringe differs from the standard auto-injectors as there is no spring-loaded needle. The syringe has a needle cap which must be pulled off to expose the needle. The needle is then inserted into the thigh and the plunger on top is pushed until it clicks.
Source:
https://fortune.com/2017/06/19/fda-mylan-epipen-adamis-symjepi/
https://www.accessdata.fda.gov/drugsatfda_docs/label/2017/207534lbl.pdf
Disclaimer: The information provided is for informational purposes only and is not intended to be a substitute for professional medical advice, diagnosis or treatment. Allergy Lifestyle Limited (t/a) Allergy Lifestyle) uses reasonable endeavours to check the accuracy of information provided however no warranty is given that they are error-free. Always seek the advice of an allergy specialist and follow your anaphylaxis emergency care plan.







I am a District nurse for a small school district in Washington. Are there trainers available for the symjepi ? I would like to train my staff before we begin to see them at school.
Hi Beth, I don’t think there are trainer pens for the Symjepi at the moment. You could try contacting the company to see when they might be available. The Symjepi should come onto the market in the first quarter of this year so it shouldn’t be too long before the trainers are available.